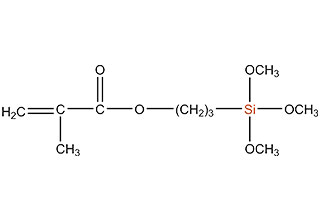
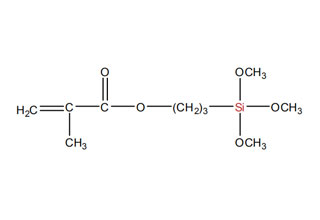
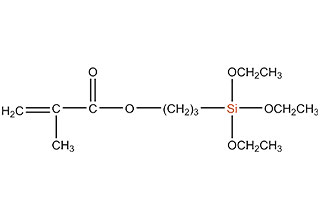
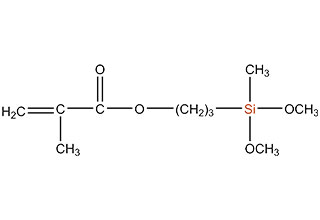
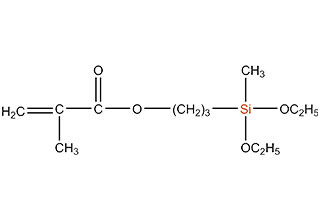
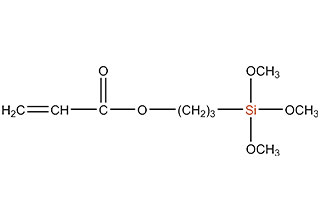
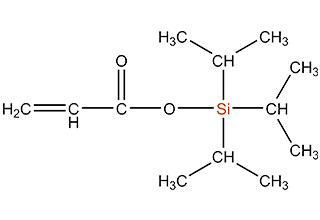
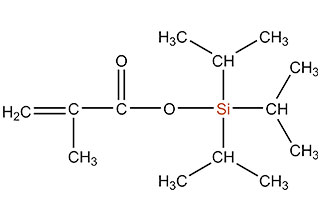
SiSiB is a worldwide leading manufacturer of organosilanes for over 30 years. SiSiB® PC4100, chemical name 3-Methacryloxypropyltrimethoxysilane, CAS No.2530-85-0, is the first and still widely used acyl silane. SiSiB® PC4100 was developed in 1989.
Acyl silane (also known as acrylate silanes, acryloxy silanes, methacrylate silanes and methacryloxy silanes) are compounds that have the silicone directly attached to the carbonyl group.
SiSiB methacryloxy silanes are bifunctional organosilanes possessing a methacryloxy group and hydrolyzable inorganic alkoxysilyl groups. The dual nature of its reactivity allows them to bind chemically to both inorganic materials and organic polymers, thus functioning as an adhesion promoter, surface modifier and as a reactant for product modification.
Methacryloxy silanes by SiSiB, a reliable silicone manufacturer, are used as surface modifier for the filler or pigment in organic resins. SiSiB methacrylate silanes chemically bind the filler or pigment into the resin offering a stable high-performance product. Gaps between the filler surface and the surrounding polymer matrix are pre-determined fracture points in the filled polymer resulting in a low mechanical stability of the product. With the treatment by silanes, gaps are removed.
SiSiB® PC4100 is a very effective coupling agent in engineered stone (also called as composite stone, breton stone, artificial stone), to improve filler dispersion, hydrophobicity, UV resistance and enhance mechanical properties.
High purity SiSiB® PC4100 MEMO silane can improve the strength of glass fiber composites and enhance the mechanical and electrical properties of many mineral-filled or reinforced composites.
SiSiB methacryloxy silanes may be copolymerized with vinyl acetate, vinyl chloride, acrylates, or methacrylate monomers to yield a moisture curable silylated polymer. The polymers can be formulated as coatings, adhesives or sealants. Adhesion and durability will be improved.
Meanwhile, acyl silanes are important intermediates of organic reaction with superior chemical and physical properties. The application of acyl silanes in organic synthesis has increased significantly over the last decade thanks to the improvement of acyl silane synthesis methods and reaction conditions.
 English
English 日本語
日本語 한국어
한국어 français
français Deutsch
Deutsch Español
Español italiano
italiano русский
русский português
português العربية
العربية tiếng việt
tiếng việt